   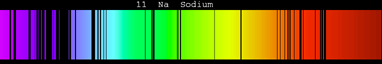
These photons have specific energy and thus a. A demonstration of the 589 nm D 2 (left) and 590 nm D 1 (right) emission sodium D lines using a wick with salt water in a flame. When they fall they release energy in the form of packets of specific energy (photons - quanta of energy). The absorption or emission spectrum would be unique for an element, and. Emission spectrum of a ceramic metal halide lamp. Although it is hard to see in this printed version, in a well-dispersed spectrum, many subtle gradations in color are visible as your eye scans from one end (violet) to the other (red). Consequently, no two elements will produce identical absorption and emission spectra. Download scientific diagram Absorption and emission spectra of DCM in methylene chloride as a function of concentration at room temperature. Students use a spectrometer, fiber optics cable, a hydrogen gas spectrum lamp, and the sun to compare emission and absorption lines, study atomic structure. When white light passes through a prism, it is dispersed and forms a continuous spectrum of all the colors. Paul Andersen explains how the photons emitted from or absorbed by an atom or nuclei is directly related to electrons moving between. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
